-
Categories
-
Pharmaceutical Intermediates
-
Active Pharmaceutical Ingredients
-
Food Additives
- Industrial Coatings
- Agrochemicals
- Dyes and Pigments
- Surfactant
- Flavors and Fragrances
- Chemical Reagents
- Catalyst and Auxiliary
- Natural Products
- Inorganic Chemistry
-
Organic Chemistry
-
Biochemical Engineering
- Analytical Chemistry
-
Cosmetic Ingredient
- Water Treatment Chemical
-
Pharmaceutical Intermediates
Promotion
ECHEMI Mall
Wholesale
Weekly Price
Exhibition
News
-
Trade Service
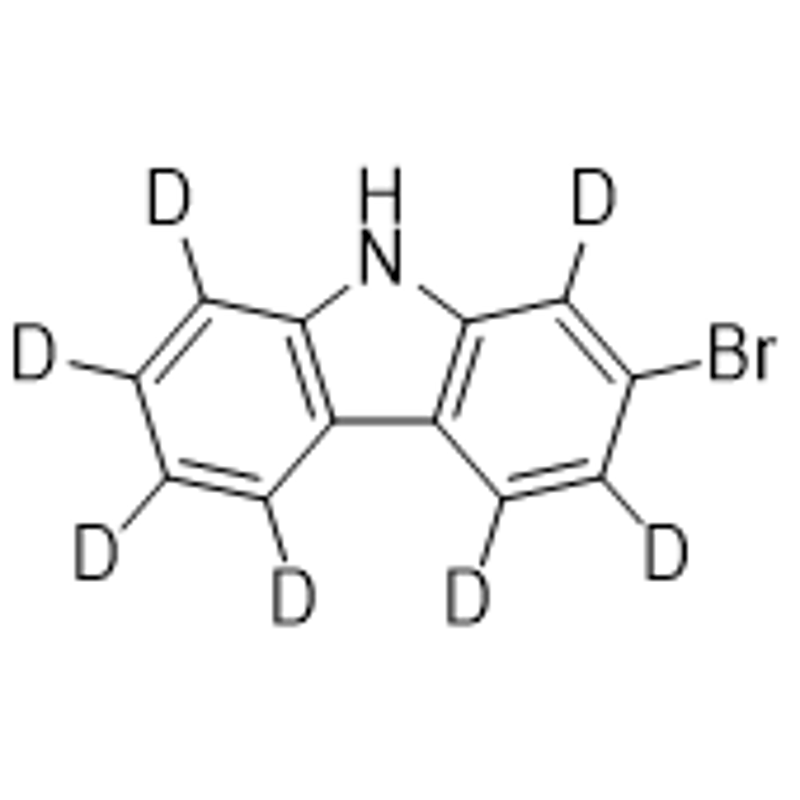
9-Bromine-11,11-dimethyl-11H-benzo[a]fluorene is a synthetic chemical compound that is widely used in various applications in the chemical industry.
The compound is synthesized using a variety of methods, each with its own advantages and disadvantages.
One of the most commonly used methods for synthesizing 9-bromine-11,11-dimethyl-11H-benzo[a]fluorene is through the Halogenation process.
In this process, 9-Bromine-11,11-dimethyl-11H-benzo[a]fluorene is treated with bromine in the presence of a solvent, resulting in the formation of the desired compound.
This method is relatively simple and inexpensive, making it a popular choice in the chemical industry.
Another method for synthesizing 9-bromine-11,11-dimethyl-11H-benzo[a]fluorene is through the Reductive Halogenation process.
In this process, the compound is treated with a reducing agent, such as hydrogen, in the presence of bromine, resulting in the formation of the desired compound.
This method is more complex and requires more advanced equipment than the Halogenation process, but it is more efficient and results in a higher yield of the desired compound.
A third method for synthesizing 9-bromine-11,11-dimethyl-11H-benzo[a]fluorene is through the Electrophilic substitution process.
In this process, the compound is treated with a reactive electrophile, such as chlorine or bromine, in the presence of a solvent, resulting in the formation of the desired compound.
This method requires the use of more specialized equipment and is more complex than the Halogenation process, but it is more versatile and can be used to synthesize a wide range of compounds.
Regardless of the method used, the synthesis of 9-bromine-11,11-dimethyl-11H-benzo[a]fluorene is an important process in the chemical industry, as the compound is widely used in the production of a variety of chemical products, such as dyes, pigments, and pharmaceuticals.
The Halogenation process is the most commonly used method, due to its simplicity and low cost, but the Reductive Halogenation and Electrophilic substitution processes are also important alternatives that are used in various applications.
In conclusion, the synthesis of 9-bromine-11,11-dimethyl-11H-benzo[a]fluorene is a critical process in the chemical industry, and there are several methods available for its synthesis, each with its own advantages and disadvantages.
The Halogenation process is the most commonly used method, but the Reductive Halogenation and Electrophilic substitution processes are also important alternatives that are used in various applications.
Regardless of the method used, the synthesis of 9-bromine-11,11-dimethyl-11H-benzo[a]fluorene is an essential part of the production process for a wide range of chemical products.