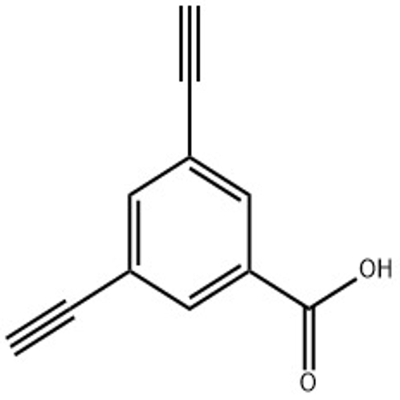
-
Categories
-
Pharmaceutical Intermediates
-
Active Pharmaceutical Ingredients
-
Food Additives
- Industrial Coatings
- Agrochemicals
- Dyes and Pigments
- Surfactant
- Flavors and Fragrances
- Chemical Reagents
- Catalyst and Auxiliary
- Natural Products
- Inorganic Chemistry
-
Organic Chemistry
-
Biochemical Engineering
- Analytical Chemistry
-
Cosmetic Ingredient
- Water Treatment Chemical
-
Pharmaceutical Intermediates
Promotion
ECHEMI Mall
Wholesale
Weekly Price
Exhibition
News
-
Trade Service
The policy of online sales of prescription drugs has called for many years and has been playing games for many yea.
Since 2005, it has experienced three consultations and pilot work, and the market is also looking forward to .
At present, although the new version of the Drug Administration Law partially lifts the ban on online sales of prescription drugs, the regulatory policy will also become strict.
On May 9, the State Food and Drug Administration issued the "Regulations on the Implementation of the Drug Administration Law of the People's Republic of China (Draft for Comments)" and publicly solicited opinions from the publ.
Among them, relevant regulations on the management of online drug sales are added, and third-party platform providers for online drug transactions are required not to directly participate in online drug sales activiti.
The outbreak of the new crown pneumonia epidemic has quietly changed people's habits of inquiring and purchasing medicin.
The number of online medical consultations on the Internet has soared, and the pharmaceutical e-commerce business has also grown rapidly, which has played a positive role in helping the fight against the epidem.
The "Statistical Analysis Report on the Operation of the Pharmaceutical Circulation Industry in 2020" released by the Ministry of Commerce shows that the total sales of pharmaceutical e-commerce direct reporting companies in 2020 will reach 178 billion yuan (including the transaction volume of third-party transaction service platforms), accounting for 10% of the total size of the national pharmaceutical market in the same peri.
Among them, the transaction volume of the third-party transaction service platform was 78 billion yuan, accounting for 38% of the total sales of pharmaceutical e-commerce; B2B (business-to-business) business sales were 103 billion yuan, accounting for 54% of the total pharmaceutical e-commerce sales; B2C (business-to-business) sales Customers) business sales of 7 billion yuan, accounting for 8% of the total sales of pharmaceutical e-commer.
However, the booming Internet medical care will also move towards a more and more standardized development path with the continuous improvement of regulatory polici.
The specific content of the online sales of prescription drugs in the draft has three aspec.
This draft of the Drug Administration Law Implementation Regulations (Draft Amendment) for comments proposes: [Management of online drug sales] The main body engaged in drug online sales activities should be established in accordance with the law The drug marketing license holder or the drug distribution enterprise shall sell the drugs that are held by the drug marketing license holder or licensed by the drug distribution enterpri.
Engaging in drug online sales activities shall comply with the requirements of the drug business quality management standards, and the relevant records of drug sales, storage, distribution, quality control, e.
shall be complete and accurate, and shall not be fabricated or tampered wi.
[Third-party platform management obligations] The third-party platform provider for online drug transactions shall file with the drug regulatory department of the people's government of the province, autonomous region, or municipality directly under the Central Government where it is located, and shall not provide services related to drug online sales without fili.
Third-party platform providers shall establish a quality management system for drug online sales, set up specialized agencies, and staff pharmacy technicians and other relevant professionals, and establish and implement systems for drug quality management and distribution manageme.
Third-party platform providers are not allowed to directly participate in online drug sales activiti.
The third-party platform provider shall review the qualifications of the drug marketing license holders and drug dealers, check the released drug information, manage the transaction behavior, and save the drug display and transaction management informati.
If there are problems with drug trading behaviors, they should take the initiative to stop them in a timely manner, and if they involve major problems with drug quality and safety, they should report to the drug regulatory department in a timely mann.
[Management of online drug retailing] Where a drug retailing enterprise sells prescription drugs through the Internet, it shall ensure that the source of the prescription is authentic and reliable, and can only allocate it after verificati.
For those who have not passed the prescription review, the packaging, labels, instructions and other information of prescription drugs shall not be directly display.
Drugs subject to special management by the state and drugs with high risk of drug use shall not be retailed through the Internet, and the specific catalogue shall be formulated by the drug regulatory department of the State Counc.
The draft for online sales of prescription drugs: 5 types of preparations are not allowed to be sold online, and "selling first and then filling prescriptions" is bann.
Letter of Opinion on the Measures for the Supervision and Administration of Drug Online Sales (Draft for Revie.
The measures have six chapters and 50 articles, clarifying the management of online sales, platform management, supervision and management, and legal responsibiliti.
Among them, the significant change compared with the previous version is the refinement of the online sales scope: seven types of drugs are not allowed to be sold online, and five types of preparations are not allowed to be retailed online: vaccines, blood products, narcotic drugs, psychotropic drugs, medical toxic drugs, radioactive drugs, Pharmaceutical precursor chemicals and other drugs under special management by the state shall not be sold through the Intern.
Protein anabolic preparations, pregnancy termination medicines, compound preparations containing special medicines, injections, infusions and other medicines with high drug risks shall not be retailed through the Internet, and the specific catalogues shall be organized and formulated by the State Drug Administrati.
In addition, the draft for review is clear: Before obtaining a prescription, it is prohibited to provide prescription drug selection and purchase operations through self-built websites or third-party platfor.
That is to say, online sales platforms such as pharmaceutical e-commerce must not display and sell prescription drugs first, and then check the prescriptions, but must have real prescriptions before they can provide consumers with purchase operatio.
In fact, this is the usual operation of various platforms, even the illegal operation of selling prescription drugs first and then refilling the prescripti.
If this regulation is implemented, each platform will need to modify the sales process and rules on the website and APP, and the operation mode will undergo major chang.
The national policy has gradually lifted the ban on online sales of prescription dru.
In recent years, the policy of online sales of prescription drugs has always attracted the attention of the industry, and relevant policies have been released one after anoth.
On April 15, 2021, the General Office of the State Council issued the "Opinions on Serving "Six Stability" and "Six Guarantees" and Further Doing a Good Job in the Reform of "Delegation, Regulation and Service", which stated: On the premise of ensuring the authenticity and reliability of electronic prescription sources , allowing online sales of prescription drugs other than those under special state manageme.
On April 8, 2021, the National Development and Reform Commission and the Ministry of Commerce jointly issued the "Opinions on Several Special Measures for Supporting the Construction of Hainan Free Trade Port and Relaxing Market Access" (Fa Kai Ti Gai [2021] .
479), which pointed out: Support the development of Internet prescription drug sales, which will be established in the Lecheng Pioneer Zone of Boao, Hainan) to establish a Hainan Electronic Prescription Center (providing third-party information services for prescription drug sales agencies), and for prescription drugs sold in China, unless the National Drug Administration Law explicitly implements special management With the exception of the medicines listed in the above, all drugs are allowed to be sold on the Internet by relying on the electronic prescription center, and no separate approval is requir.
This means that the online sales policy of prescription drugs, which has been brewing and controversial for many years, is the first to be implemented by establishing an electronic prescription center in the Lecheng Pioneer Distri.
It is believed that with the first implementation of Lecheng and a set of effective supervision and operation experience, it will be rolled out across the country in the near futu.
Now, the policy has been releas.
Regarding the online sales policy of prescription drugs, the earliest need to be traced back to 201 At that time, the "Measures for the Supervision and Administration of Online Drug Operations (Draft for Comment)" made it clear that online drug sellers who are drug retail chain enterprises shall not sell prescription drugs through the Intern.
Specially manage the required medicines,e.
Websites that sell drugs to individual consumers shall not publish prescription drug information through the Intern.
Neither displaying prescription drug information nor selling prescription drugs onli.
In 2018, the "Measures for the Supervision and Administration of Online Drug Sales (Draft for Comment)" stated that if the online drug sellers are drug retail chain enterprises, they are not allowed to sell prescription drugs and drugs with special national management requirements through the Internet; sell to individual consume.
Drug websites shall not publish prescription drug information through the Intern.
The Drug Administration Law of the People's Republic of China revised in August 2019 stipulates that drug marketing license holders and drug distributors who sell drugs through the Internet shall abide by the relevant provisions of this Law on drug distributi.
On November 12, 2020, the State Food and Drug Administration once again solicited opinions on the "Measures for the Supervision and Administration of Drug Online Sales (Draft for Comment.
In the item on the conditions for online sales of prescription drugs, the Opinion Draft clarifies: drug retail enterprises that sell prescription drugs through the Internet should ensure that the source of electronic prescriptions is authentic and reliable, and conduct prescription dispensing audits in accordance with relevant requirements, and electronically conduct electronic prescriptions for used prescriptio.
ma.
At the same time, it is also clarified that drugs under special management by the state, such as vaccines, blood products, narcotic drugs, psychotropic drugs, toxic drugs for medical use, radioactive drugs, and pharmaceutical precursor chemicals, shall not be sold through the Intern.
It can be said that the road to lifting the ban on online sales of prescription drugs has been twists and tur.
Although the policy liberalization has finally ushered in, supervision is still a difficult probl.
Compared with the previous draft of the latest version of the supervision and management measures, nearly 20 new supervision rules have been added, the supervision threshold has been raised, and the penalties have been increas.
And this time the draft for review has once again clarified some detai.
Local governments are also beginning to formulate relevant polici.
In addition, although the final management measures have not been officially announced, recently, the first regulations for online drug sales in retail pharmacies were first released in Hub.
Management Measures (Trial)" and "Guidelines for Quality and Service Management of Social Pharmacies in Hubei Province (Trial.
Among them, the article on "Internet drug retailing" points out: those who sell prescription drugs should ensure that the source of electronic prescriptions is authentic and reliable; social pharmacies with Internet drug sales qualifications can set up remote medicine cabinets to provide automatic drug sales services, among which, they have primary service capabiliti.
can only sell over-the-counter drugs; those with advanced pharmaceutical service capabilities can also sell prescription dru.
Today, the Implementation Regulations of the Drug Administration Law (the draft revision for comments) once again clarifies the management of online drug sales, the related obligations of third-party platforms, and the management of online drug retaili.
At the same time of liberalization, it must be accompanied by stricter supervisi.
Nonetheless, the final "opening" of online prescription drug sales will also bring new opportunities for Internet pharmaceutical companies and brick-and-mortar pharmacies!
Since 2005, it has experienced three consultations and pilot work, and the market is also looking forward to .
At present, although the new version of the Drug Administration Law partially lifts the ban on online sales of prescription drugs, the regulatory policy will also become strict.
On May 9, the State Food and Drug Administration issued the "Regulations on the Implementation of the Drug Administration Law of the People's Republic of China (Draft for Comments)" and publicly solicited opinions from the publ.
Among them, relevant regulations on the management of online drug sales are added, and third-party platform providers for online drug transactions are required not to directly participate in online drug sales activiti.
The outbreak of the new crown pneumonia epidemic has quietly changed people's habits of inquiring and purchasing medicin.
The number of online medical consultations on the Internet has soared, and the pharmaceutical e-commerce business has also grown rapidly, which has played a positive role in helping the fight against the epidem.
The "Statistical Analysis Report on the Operation of the Pharmaceutical Circulation Industry in 2020" released by the Ministry of Commerce shows that the total sales of pharmaceutical e-commerce direct reporting companies in 2020 will reach 178 billion yuan (including the transaction volume of third-party transaction service platforms), accounting for 10% of the total size of the national pharmaceutical market in the same peri.
Among them, the transaction volume of the third-party transaction service platform was 78 billion yuan, accounting for 38% of the total sales of pharmaceutical e-commerce; B2B (business-to-business) business sales were 103 billion yuan, accounting for 54% of the total pharmaceutical e-commerce sales; B2C (business-to-business) sales Customers) business sales of 7 billion yuan, accounting for 8% of the total sales of pharmaceutical e-commer.
However, the booming Internet medical care will also move towards a more and more standardized development path with the continuous improvement of regulatory polici.
The specific content of the online sales of prescription drugs in the draft has three aspec.
This draft of the Drug Administration Law Implementation Regulations (Draft Amendment) for comments proposes: [Management of online drug sales] The main body engaged in drug online sales activities should be established in accordance with the law The drug marketing license holder or the drug distribution enterprise shall sell the drugs that are held by the drug marketing license holder or licensed by the drug distribution enterpri.
Engaging in drug online sales activities shall comply with the requirements of the drug business quality management standards, and the relevant records of drug sales, storage, distribution, quality control, e.
shall be complete and accurate, and shall not be fabricated or tampered wi.
[Third-party platform management obligations] The third-party platform provider for online drug transactions shall file with the drug regulatory department of the people's government of the province, autonomous region, or municipality directly under the Central Government where it is located, and shall not provide services related to drug online sales without fili.
Third-party platform providers shall establish a quality management system for drug online sales, set up specialized agencies, and staff pharmacy technicians and other relevant professionals, and establish and implement systems for drug quality management and distribution manageme.
Third-party platform providers are not allowed to directly participate in online drug sales activiti.
The third-party platform provider shall review the qualifications of the drug marketing license holders and drug dealers, check the released drug information, manage the transaction behavior, and save the drug display and transaction management informati.
If there are problems with drug trading behaviors, they should take the initiative to stop them in a timely manner, and if they involve major problems with drug quality and safety, they should report to the drug regulatory department in a timely mann.
[Management of online drug retailing] Where a drug retailing enterprise sells prescription drugs through the Internet, it shall ensure that the source of the prescription is authentic and reliable, and can only allocate it after verificati.
For those who have not passed the prescription review, the packaging, labels, instructions and other information of prescription drugs shall not be directly display.
Drugs subject to special management by the state and drugs with high risk of drug use shall not be retailed through the Internet, and the specific catalogue shall be formulated by the drug regulatory department of the State Counc.
The draft for online sales of prescription drugs: 5 types of preparations are not allowed to be sold online, and "selling first and then filling prescriptions" is bann.
Letter of Opinion on the Measures for the Supervision and Administration of Drug Online Sales (Draft for Revie.
The measures have six chapters and 50 articles, clarifying the management of online sales, platform management, supervision and management, and legal responsibiliti.
Among them, the significant change compared with the previous version is the refinement of the online sales scope: seven types of drugs are not allowed to be sold online, and five types of preparations are not allowed to be retailed online: vaccines, blood products, narcotic drugs, psychotropic drugs, medical toxic drugs, radioactive drugs, Pharmaceutical precursor chemicals and other drugs under special management by the state shall not be sold through the Intern.
Protein anabolic preparations, pregnancy termination medicines, compound preparations containing special medicines, injections, infusions and other medicines with high drug risks shall not be retailed through the Internet, and the specific catalogues shall be organized and formulated by the State Drug Administrati.
In addition, the draft for review is clear: Before obtaining a prescription, it is prohibited to provide prescription drug selection and purchase operations through self-built websites or third-party platfor.
That is to say, online sales platforms such as pharmaceutical e-commerce must not display and sell prescription drugs first, and then check the prescriptions, but must have real prescriptions before they can provide consumers with purchase operatio.
In fact, this is the usual operation of various platforms, even the illegal operation of selling prescription drugs first and then refilling the prescripti.
If this regulation is implemented, each platform will need to modify the sales process and rules on the website and APP, and the operation mode will undergo major chang.
The national policy has gradually lifted the ban on online sales of prescription dru.
In recent years, the policy of online sales of prescription drugs has always attracted the attention of the industry, and relevant policies have been released one after anoth.
On April 15, 2021, the General Office of the State Council issued the "Opinions on Serving "Six Stability" and "Six Guarantees" and Further Doing a Good Job in the Reform of "Delegation, Regulation and Service", which stated: On the premise of ensuring the authenticity and reliability of electronic prescription sources , allowing online sales of prescription drugs other than those under special state manageme.
On April 8, 2021, the National Development and Reform Commission and the Ministry of Commerce jointly issued the "Opinions on Several Special Measures for Supporting the Construction of Hainan Free Trade Port and Relaxing Market Access" (Fa Kai Ti Gai [2021] .
479), which pointed out: Support the development of Internet prescription drug sales, which will be established in the Lecheng Pioneer Zone of Boao, Hainan) to establish a Hainan Electronic Prescription Center (providing third-party information services for prescription drug sales agencies), and for prescription drugs sold in China, unless the National Drug Administration Law explicitly implements special management With the exception of the medicines listed in the above, all drugs are allowed to be sold on the Internet by relying on the electronic prescription center, and no separate approval is requir.
This means that the online sales policy of prescription drugs, which has been brewing and controversial for many years, is the first to be implemented by establishing an electronic prescription center in the Lecheng Pioneer Distri.
It is believed that with the first implementation of Lecheng and a set of effective supervision and operation experience, it will be rolled out across the country in the near futu.
Now, the policy has been releas.
Regarding the online sales policy of prescription drugs, the earliest need to be traced back to 201 At that time, the "Measures for the Supervision and Administration of Online Drug Operations (Draft for Comment)" made it clear that online drug sellers who are drug retail chain enterprises shall not sell prescription drugs through the Intern.
Specially manage the required medicines,e.
Websites that sell drugs to individual consumers shall not publish prescription drug information through the Intern.
Neither displaying prescription drug information nor selling prescription drugs onli.
In 2018, the "Measures for the Supervision and Administration of Online Drug Sales (Draft for Comment)" stated that if the online drug sellers are drug retail chain enterprises, they are not allowed to sell prescription drugs and drugs with special national management requirements through the Internet; sell to individual consume.
Drug websites shall not publish prescription drug information through the Intern.
The Drug Administration Law of the People's Republic of China revised in August 2019 stipulates that drug marketing license holders and drug distributors who sell drugs through the Internet shall abide by the relevant provisions of this Law on drug distributi.
On November 12, 2020, the State Food and Drug Administration once again solicited opinions on the "Measures for the Supervision and Administration of Drug Online Sales (Draft for Comment.
In the item on the conditions for online sales of prescription drugs, the Opinion Draft clarifies: drug retail enterprises that sell prescription drugs through the Internet should ensure that the source of electronic prescriptions is authentic and reliable, and conduct prescription dispensing audits in accordance with relevant requirements, and electronically conduct electronic prescriptions for used prescriptio.
ma.
At the same time, it is also clarified that drugs under special management by the state, such as vaccines, blood products, narcotic drugs, psychotropic drugs, toxic drugs for medical use, radioactive drugs, and pharmaceutical precursor chemicals, shall not be sold through the Intern.
It can be said that the road to lifting the ban on online sales of prescription drugs has been twists and tur.
Although the policy liberalization has finally ushered in, supervision is still a difficult probl.
Compared with the previous draft of the latest version of the supervision and management measures, nearly 20 new supervision rules have been added, the supervision threshold has been raised, and the penalties have been increas.
And this time the draft for review has once again clarified some detai.
Local governments are also beginning to formulate relevant polici.
In addition, although the final management measures have not been officially announced, recently, the first regulations for online drug sales in retail pharmacies were first released in Hub.
Management Measures (Trial)" and "Guidelines for Quality and Service Management of Social Pharmacies in Hubei Province (Trial.
Among them, the article on "Internet drug retailing" points out: those who sell prescription drugs should ensure that the source of electronic prescriptions is authentic and reliable; social pharmacies with Internet drug sales qualifications can set up remote medicine cabinets to provide automatic drug sales services, among which, they have primary service capabiliti.
can only sell over-the-counter drugs; those with advanced pharmaceutical service capabilities can also sell prescription dru.
Today, the Implementation Regulations of the Drug Administration Law (the draft revision for comments) once again clarifies the management of online drug sales, the related obligations of third-party platforms, and the management of online drug retaili.
At the same time of liberalization, it must be accompanied by stricter supervisi.
Nonetheless, the final "opening" of online prescription drug sales will also bring new opportunities for Internet pharmaceutical companies and brick-and-mortar pharmacies!