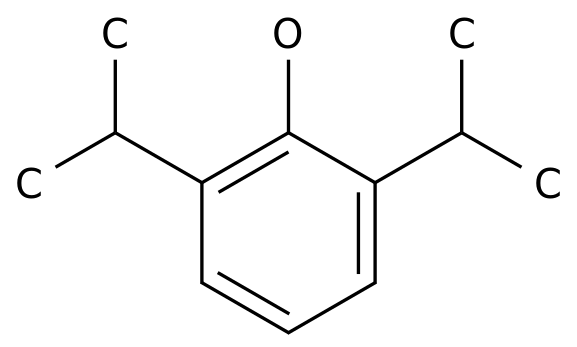
-
Categories
-
Pharmaceutical Intermediates
-
Active Pharmaceutical Ingredients
-
Food Additives
- Industrial Coatings
- Agrochemicals
- Dyes and Pigments
- Surfactant
- Flavors and Fragrances
- Chemical Reagents
- Catalyst and Auxiliary
- Natural Products
- Inorganic Chemistry
-
Organic Chemistry
-
Biochemical Engineering
- Analytical Chemistry
-
Cosmetic Ingredient
- Water Treatment Chemical
-
Pharmaceutical Intermediates
Promotion
ECHEMI Mall
Wholesale
Weekly Price
Exhibition
News
-
Trade Service
[March 5, 2021 / Medical Information List] Eli Lilly's tirzepatide head-to-head phase III study was successful; Amgen acquired FivePrime for US$1.
[March 5, 2021 / List of Medical Information] Daily news about fresh medicines and medical news, Speed Reading Club will pay attention to you together!
Part1 Policy Brief
Part1 policy briefing Part1 policy briefing2021 National Two Sessions: The per capita financial subsidy standard for resident medical insurance will be increased by 30 yuan
2021 National Two Sessions: The per capita financial subsidy standard for resident medical insurance will be increased by 30 yuanAt the fourth meeting of the 13th National People’s Congress held on the morning of the 5th, Premier Li Keqiang pointed out in his report on the work of the government that the per capita financial subsidy standard for residents’ medical insurance and basic public health service funds will be increased by 30 yuan and 5 yuan, respectively, to promote basic Medical insurance is coordinated at the provincial level, and outpatient expenses are directly settled across provinces.
Government work report: Incorporate more chronic disease, common disease drugs and high-value consumables into centralized procurement
Government work report: Incorporate more chronic disease, common disease drugs and high-value consumables into centralized procurementOn the morning of March 5, the fourth session of the 13th National People's Congress opened.
A pharmaceutical company was warned for failing to comply with GMP during the inspection process
A pharmaceutical company was warned for failing to comply with GMP during the inspection processOn March 4, the Fujian Provincial Food and Drug Administration issued an administrative penalty information disclosure form, which showed that Fujian Minghua Pharmaceutical violated the "Pharmaceutical Production Quality Management Standards" due to its failure to comply with GMP regulations during the on-site inspection process and product inspection process during the certificate renewal process.
Part2 Observation
Part2 Sankei Observation Part2 Sankei ObservationAmgen acquires FivePrime for US$1.
Amgen acquires FivePrime for US$1.
Novartis dissolves sales team for oxcarbazepine tablets
Novartis dissolves sales team for oxcarbazepine tabletsRecently, news broke that the sales team for oxcarbazepine tablets of Novartis’s CNS product line has been disbanded.
Sigma Biotech's angel round completes about 60 million yuan in financing
Sigma Biotech's angel round completes about 60 million yuan in financingArtery Network learned for the first time that, recently, Sigma Biosciences announced that it has over-completed an angel round of financing of approximately RMB 60 million.
Wuhan Zhihua Technology completes a round of financing of over 30 million yuan
Wuhan Zhihua Technology completes a round of financing of over 30 million yuanArtery Network learned for the first time that Wuhan Zhihua Technology recently announced the completion of a series A financing of more than 30 million yuan.
Exscientia announces the completion of a US$100 million Series C financing
Exscientia announces the completion of a US$100 million Series C financingRecently, Exscientia announced that a fund managed by BlackRock has participated in the company's Series C investment.
Part3 Medicine News
Part3 Medicine News Part3 Medicine NewsEli Lilly’s tirzepatide head-to-head phase III study succeeds
Eli Lilly’s tirzepatide head-to-head phase III study succeedsOn March 4, Eli Lilly announced the results of a 40-week phase 3 clinical trial of SURPASS-2.
Youhe Pharmaceutical announced that its CTLA-4 combined with Junshi Bio-PD-1 Phase I clinical results have achieved breakthrough results
Youhe Pharmaceutical announced that its CTLA-4 combined with Junshi Bio-PD-1 Phase I clinical results have achieved breakthrough resultsYouhe Pharmaceutical announced on March 4 that its anti-CTLA-4 antibody combined with Junshi Bio's anti-PD-1 monoclonal antibody drug teriprizumab injection showed positive anti-tumor activity in the dose escalation phase of the Australian Phase I clinical study .
Galapagos releases interim clinical safety data of filgotinib
Galapagos releases interim clinical safety data of filgotinibOn March 4, Galapagos NV, a scientific partner of Gilead, announced the interim results from two studies MANTA and MANTA-RAy.
Biotech terminates clinical development of PD-1 monoclonal antibody and Trop2 antibody conjugate drug project
Biotech terminates clinical development of PD-1 monoclonal antibody and Trop2 antibody conjugate drug projectOn the 4th, Biotech issued an announcement stating that the company decided to terminate its research and development of BAT800 (recombinant humanized anti-Trop2 monoclonal antibody-maytansine conjugate for injection) and Clinical trial of BAT1306 (namely recombinant humanized anti-PD-1 monoclonal antibody injection) project.
Renhe Yikang Pharmaceutical Dexmedetomidine Hydrochloride Injection Approved
Renhe Yikang Pharmaceutical Dexmedetomidine Hydrochloride Injection ApprovedOn March 4, the official website of CDE showed that Hebei Renhe Yikang Pharmaceutical's dexmedetomidine hydrochloride injection was accepted as imitation 3 types.
The supplementary application for vinorelbine tartrate injection of Hausen Pharmaceuticals was approved and reviewed
The supplementary application for vinorelbine tartrate injection of Hausen Pharmaceuticals was approved and reviewedOn March 3, the official website of the State Food and Drug Administration showed that Jiangsu Haosen Pharmaceutical's vinorelbine tartrate injection was approved as a supplementary application.
Junshi Bio: JS103 injection received the notification of acceptance of drug clinical trial application
Junshi Bio: JS103 injection received the notification of acceptance of drug clinical trial application
On March 5, Junshi Bio announced that the company received the "Notice of Acceptance" approved and issued by the National Medical Products Administration, and the clinical trial application for JS103 injection was accepted.
JS103 is a pegylated uricase derivative independently developed by the company, which is mainly used for the treatment of hyperuricemia with or without gout.
(Junshi biological announcement)
Liantuo Biosciences Introduces Anti-RSV Drug Candidates for More than 100 Million U.
S.
Dollars
S.
Dollars
Recently, Liantuo Biotechnology announced that it has reached an exclusive cooperation and licensing agreement with ReViral, and will jointly develop and commercialize ReViral's drug candidate sisunatovir in mainland China, Hong Kong, Macau, and Singapore.
This is a small molecule drug for the treatment of respiratory syncytial virus infection.
It has been approved by the FDA for fast-track entry.
It is currently undergoing phase 2 clinical trials in children and adults with immunocompromised patients.
(WuXi AppTec)