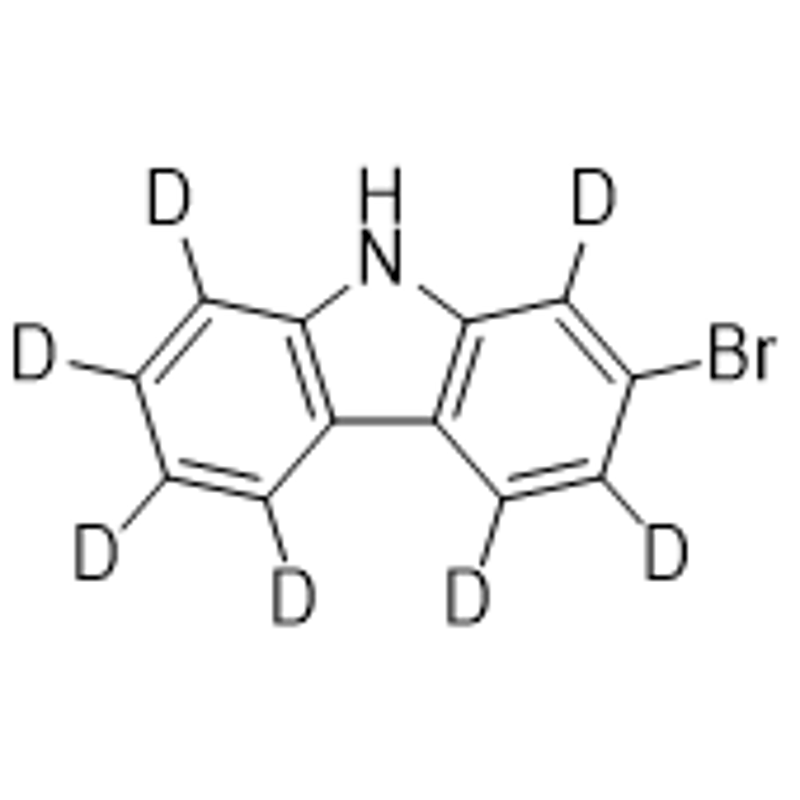
-
Categories
-
Pharmaceutical Intermediates
-
Active Pharmaceutical Ingredients
-
Food Additives
- Industrial Coatings
- Agrochemicals
- Dyes and Pigments
- Surfactant
- Flavors and Fragrances
- Chemical Reagents
- Catalyst and Auxiliary
- Natural Products
- Inorganic Chemistry
-
Organic Chemistry
-
Biochemical Engineering
- Analytical Chemistry
-
Cosmetic Ingredient
- Water Treatment Chemical
-
Pharmaceutical Intermediates
Promotion
ECHEMI Mall
Wholesale
Weekly Price
Exhibition
News
-
Trade Service
Carrying out drug price correction and price limit listing is conducive to solving the falsely high drug prices and rectifying the order of the pharmaceutical market
.
Since the beginning of this year, many places including Hunan and Sichuan have posted lists of drug withdrawals from the Internet, involving hundreds of products and dozens of pharmaceutical companies
.
Recently, a notice issued by the Shaanxi Provincial Public Resource Trading Center on the adjustment of the product listing catalogue for the dynamic adjustment of pharmaceutical products on the Internet shows that a total of 57 drugs have been adjusted this time, of which 7 are included in the direct listing catalogue, and the listings are revoked.
There are 50 of them
.
Among the 50 withdrawal drugs, there are many commonly used drugs such as Vitamin C Yinqiao Tablets, Banlangen Injection, Cefmetazole Sodium for Injection, Ganmao Qingre Granules, Mezlocillin Sodium Sulbactam Sodium for Injection, Yinhuang Oral Liquid, etc.
Cefmetazole sodium, omeprazole sodium for injection and other large varieties of over 1 billion yuan, involving Qianyuan Pharmaceutical, Minkang Pharmaceutical, Luoxin Pharmaceutical and other pharmaceutical companies
.
It is worth mentioning that this time Minkang Pharmaceutical has a total of 23 drug applications and withdrawals, including Ammonia Huangmin Capsules, Shengui Yangxue Oral Liquid, Jinming Hewei Capsules, Vitamin C Yinqiao Tablets, Yinhuang Oral Liquid, etc.
Chinese patent medicine varieties
.
In addition to Shaanxi, Minkang Pharmaceutical was also included in the "Notice on Publicizing Some Enterprises' Application for Revocation of Platform-Based Purchasing Qualifications" issued by Shanxi Provincial Pharmaceutical Equipment Centralized Bidding and Purchasing Center on November 23, 2021.
There are 8 manufacturers involved in 32 drugs, of which Minkang Pharmaceutical still has a high proportion of drugs withdrawn from the network, with 17 varieties withdrawn from the network, accounting for 53.
13%
.
In August 2021, the Heilongjiang Provincial Drug Centralized Purchasing Network issued the "Notice on the withdrawal of some drugs from the provincial procurement platform", among which Tianwang Buxin Pill of Hubei Minkang Pharmaceuticals suspended the production of this product due to sales and operation adjustments
.
Some analysts believe that there are two main reasons for the decision of pharmaceutical companies to withdraw from the network.
First, the market share of these companies’ products in the local market where they are withdrawn from the network is not large; The supply is directly related, and only the decision to withdraw from the network can be made
.
From the perspective of Chinese patent medicine companies, rising production costs have brought challenges to the performance of related companies
.
Since 2020, the prices of many bulk raw materials have continued to rise, and even rare doublings have occurred..
In the process of rising prices of Chinese herbal medicines, the raw materials produced by Chinese patent medicine enterprises cannot be stabilized and guaranteed, which directly affects the performance of enterprises
.
In addition, the production cost is also directly related to the product quality of Chinese patent medicine enterprises
.
The industry pointed out that because the company's income is directly related to the profit of the product, in the current environment where raw materials are generally rising, the winning company may lose money to perform the contract in order to meet the requirements of quality and quantity supply, or have to purchase raw materials with lower prices to match.
The winning bid price and the quality of the raw materials purchased may decline, resulting in a decline in corporate reputation and brand influence
.
The industry expects that with the advancement of the centralized procurement of proprietary Chinese medicines, the entire industry will face a major reshuffle in the future, and products with smaller market scales and no special dosage forms will be eliminated at an accelerated pace
.
It is understood that compared with 2019, the market share of Chinese patent medicines is obviously showing a downward trend, and the future will decline or even worse
.
It is worth mentioning that, according to the "Implementation Plan for the Dynamic Adjustment of Drug Sunshine Hanging on the Internet" issued by the Shaanxi Provincial Public Resource Trading Center in July 2020, after a manufacturer applies for the cancellation of a product on the Internet, the cancellation will not be accepted within two years from the date of cancellation.
Online application for products
.
Disclaimer: Under no circumstances does the information or opinions expressed in this article constitute investment advice to anyone
.
.
Since the beginning of this year, many places including Hunan and Sichuan have posted lists of drug withdrawals from the Internet, involving hundreds of products and dozens of pharmaceutical companies
.
Recently, a notice issued by the Shaanxi Provincial Public Resource Trading Center on the adjustment of the product listing catalogue for the dynamic adjustment of pharmaceutical products on the Internet shows that a total of 57 drugs have been adjusted this time, of which 7 are included in the direct listing catalogue, and the listings are revoked.
There are 50 of them
.
Among the 50 withdrawal drugs, there are many commonly used drugs such as Vitamin C Yinqiao Tablets, Banlangen Injection, Cefmetazole Sodium for Injection, Ganmao Qingre Granules, Mezlocillin Sodium Sulbactam Sodium for Injection, Yinhuang Oral Liquid, etc.
Cefmetazole sodium, omeprazole sodium for injection and other large varieties of over 1 billion yuan, involving Qianyuan Pharmaceutical, Minkang Pharmaceutical, Luoxin Pharmaceutical and other pharmaceutical companies
.
It is worth mentioning that this time Minkang Pharmaceutical has a total of 23 drug applications and withdrawals, including Ammonia Huangmin Capsules, Shengui Yangxue Oral Liquid, Jinming Hewei Capsules, Vitamin C Yinqiao Tablets, Yinhuang Oral Liquid, etc.
Chinese patent medicine varieties
.
In addition to Shaanxi, Minkang Pharmaceutical was also included in the "Notice on Publicizing Some Enterprises' Application for Revocation of Platform-Based Purchasing Qualifications" issued by Shanxi Provincial Pharmaceutical Equipment Centralized Bidding and Purchasing Center on November 23, 2021.
There are 8 manufacturers involved in 32 drugs, of which Minkang Pharmaceutical still has a high proportion of drugs withdrawn from the network, with 17 varieties withdrawn from the network, accounting for 53.
13%
.
In August 2021, the Heilongjiang Provincial Drug Centralized Purchasing Network issued the "Notice on the withdrawal of some drugs from the provincial procurement platform", among which Tianwang Buxin Pill of Hubei Minkang Pharmaceuticals suspended the production of this product due to sales and operation adjustments
.
Some analysts believe that there are two main reasons for the decision of pharmaceutical companies to withdraw from the network.
First, the market share of these companies’ products in the local market where they are withdrawn from the network is not large; The supply is directly related, and only the decision to withdraw from the network can be made
.
From the perspective of Chinese patent medicine companies, rising production costs have brought challenges to the performance of related companies
.
Since 2020, the prices of many bulk raw materials have continued to rise, and even rare doublings have occurred..
In the process of rising prices of Chinese herbal medicines, the raw materials produced by Chinese patent medicine enterprises cannot be stabilized and guaranteed, which directly affects the performance of enterprises
.
In addition, the production cost is also directly related to the product quality of Chinese patent medicine enterprises
.
The industry pointed out that because the company's income is directly related to the profit of the product, in the current environment where raw materials are generally rising, the winning company may lose money to perform the contract in order to meet the requirements of quality and quantity supply, or have to purchase raw materials with lower prices to match.
The winning bid price and the quality of the raw materials purchased may decline, resulting in a decline in corporate reputation and brand influence
.
The industry expects that with the advancement of the centralized procurement of proprietary Chinese medicines, the entire industry will face a major reshuffle in the future, and products with smaller market scales and no special dosage forms will be eliminated at an accelerated pace
.
It is understood that compared with 2019, the market share of Chinese patent medicines is obviously showing a downward trend, and the future will decline or even worse
.
It is worth mentioning that, according to the "Implementation Plan for the Dynamic Adjustment of Drug Sunshine Hanging on the Internet" issued by the Shaanxi Provincial Public Resource Trading Center in July 2020, after a manufacturer applies for the cancellation of a product on the Internet, the cancellation will not be accepted within two years from the date of cancellation.
Online application for products
.
Disclaimer: Under no circumstances does the information or opinions expressed in this article constitute investment advice to anyone
.