-
Categories
-
Pharmaceutical Intermediates
-
Active Pharmaceutical Ingredients
-
Food Additives
- Industrial Coatings
- Agrochemicals
- Dyes and Pigments
- Surfactant
- Flavors and Fragrances
- Chemical Reagents
- Catalyst and Auxiliary
- Natural Products
- Inorganic Chemistry
-
Organic Chemistry
-
Biochemical Engineering
- Analytical Chemistry
-
Cosmetic Ingredient
- Water Treatment Chemical
-
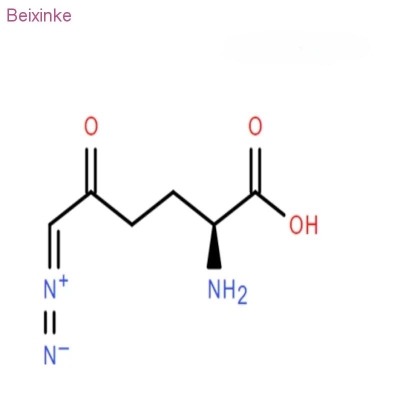
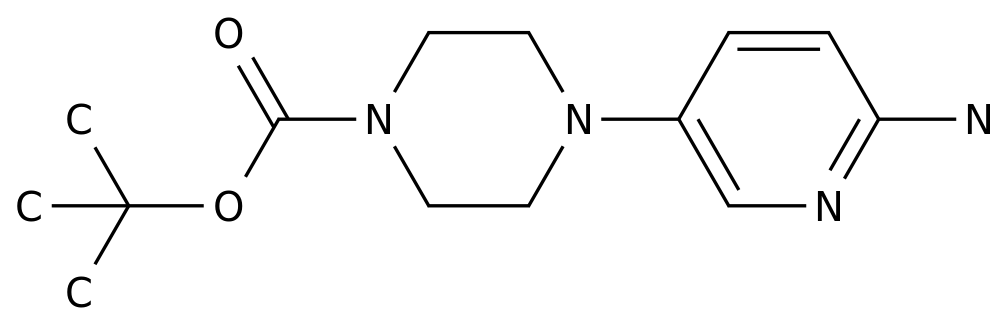
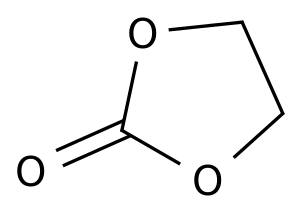
Pharmaceutical Intermediates
Promotion
ECHEMI Mall
Wholesale
Weekly Price
Exhibition
News
-
Trade Service
According to the news of the Nansha District Market Supervision Bureau of Guangzhou on the 5th, the district has continued to build an industrial development "fusion agent" in recent years, focusing on building a new model of production and marketing of Hong Kong and Macao pharmaceutical devices, actively promoting the "reform of the drug marketing authorization holder system in the Guangdong-Hong Kong-Macao Greater Bay Area", and opening up a "green channel"
for Hong Kong and Macao enterprises that meet relevant conditions to develop in Nansha.
for Hong Kong and Macao enterprises that meet relevant conditions to develop in Nansha.
According to the Nansha District Market Supervision Bureau, in order to build a biomedical industry cluster, give play to the main role of enterprise innovation, and promote the transformation of R&D results in Nansha, the bureau actively piloted the early intervention and guidance of biomedical enterprises, established a "green channel", and explored the establishment of drug and medical device evaluation and inspection service institutions in the jurisdiction to provide full-chain services
for enterprise license declaration, registration and filing consultation.
for enterprise license declaration, registration and filing consultation.
The relevant person in charge of the Nansha District Market Supervision Bureau said that the bureau is taking many measures to promote "the reform of the drug marketing authorization holder system in the Guangdong-Hong Kong-Macao Greater Bay Area" and actively strengthen the docking
with Hong Kong and Macao enterprises.
with Hong Kong and Macao enterprises.
The person in charge introduced that the bureau promoted the Hong Kong Lepu Pharmaceutical Company's lidocaine hydrochloride powder needleless percutaneous injection drug delivery system Zingo? Jinlaiyi commissioned production in Nansha Zhaoke Pharmaceutical (Guangzhou) Co.
, Ltd.
; Support the pilot of cross-border e-commerce import of OTC (over-the-counter) drugs in Hong Kong and Macao, and rely on chain stores to promote the promotion of Class B OTC drug counters in Nansha; It also carried out strategic cooperation with China Resources Guangdong Pharmaceutical Co.
, Ltd.
, a designated enterprise for logistics and distribution of "Hong Kong and Macao Pharmaceutical Equipment Connect", to establish a new model
of drug storage and distribution in pilot medical institutions.
, Ltd.
; Support the pilot of cross-border e-commerce import of OTC (over-the-counter) drugs in Hong Kong and Macao, and rely on chain stores to promote the promotion of Class B OTC drug counters in Nansha; It also carried out strategic cooperation with China Resources Guangdong Pharmaceutical Co.
, Ltd.
, a designated enterprise for logistics and distribution of "Hong Kong and Macao Pharmaceutical Equipment Connect", to establish a new model
of drug storage and distribution in pilot medical institutions.
In addition, in order to solve the key problems of enterprise drug R&D, Nansha District has also actively promoted the pilot of the "white list" system for the import of pharmaceutical R&D items, and combined with the "Several Measures of the General Administration of Customs to Support Guangzhou Nansha to Deepen Guangdong-Hong Kong-Macao Comprehensive Cooperation Facing the World", solicit the needs of enterprises through multiple channels, strengthen research in conjunction with customs and other departments, and continue to explore measures to improve the import facilitation of
biomedical R&D items.
(End)
biomedical R&D items.
(End)