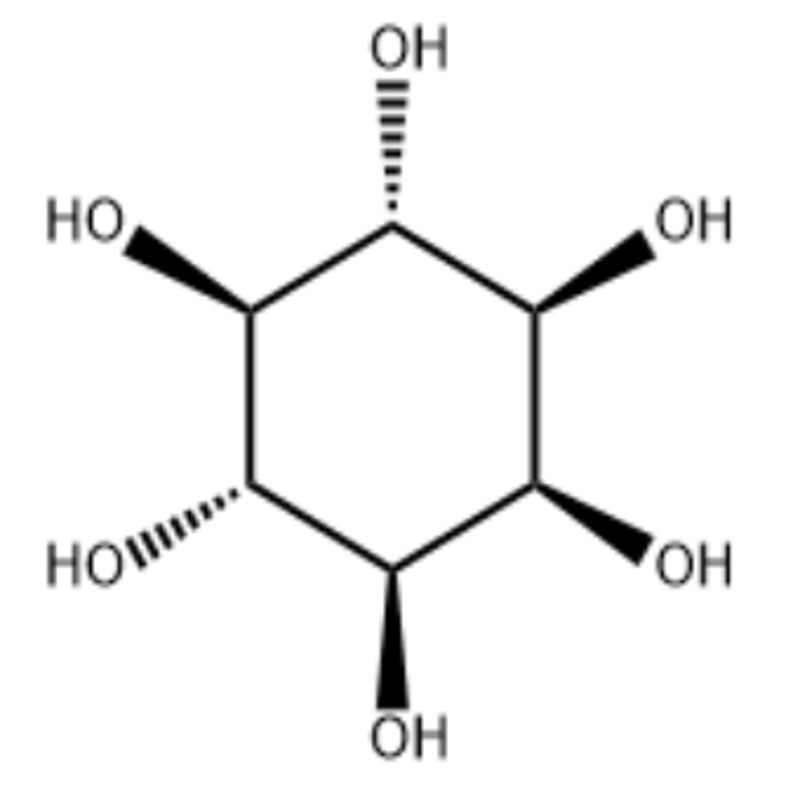
-
Categories
-
Pharmaceutical Intermediates
-
Active Pharmaceutical Ingredients
-
Food Additives
- Industrial Coatings
- Agrochemicals
- Dyes and Pigments
- Surfactant
- Flavors and Fragrances
- Chemical Reagents
- Catalyst and Auxiliary
- Natural Products
- Inorganic Chemistry
-
Organic Chemistry
-
Biochemical Engineering
- Analytical Chemistry
-
Cosmetic Ingredient
- Water Treatment Chemical
-
Pharmaceutical Intermediates
Promotion
ECHEMI Mall
Wholesale
Weekly Price
Exhibition
News
-
Trade Service
Guide This guide is used by health departments and disease control agencies at all levels to guide vaccination units to carry out new coronavirus vaccination.
The guide mentions that people with chronic diseases who are well controlled by drugs are not considered contraindicated for new coronavirus vaccination and are recommended to be vaccinated.
Source: National Health Commission's official website.
The National Health Commission's website reported on March 29 that at present, my country has 5 manufacturers of new coronavirus vaccines approved for conditional listing or emergency use.
The results of the interim analysis of the phase III clinical trials of the three inactivated vaccines and adenovirus vector vaccines that were conditionally approved for marketing showed that the protective efficacy of the vaccines all met the guidelines for the clinical evaluation of new coronavirus preventive vaccines (trial) by the National Medical Products Administration.
The requirements also meet the index requirements recommended by the World Health Organization’s "New Coronavirus Vaccine Target Product Characteristics".
Surveillance data of suspected abnormal response to vaccination after large-scale vaccination in key populations during clinical trials and emergency use phases and earlier stages show that the new coronavirus vaccine is safe.
The results of the Phase II clinical trial of the recombinant new coronavirus vaccine (CHO cell) approved for emergency use showed good immunogenicity and safety.
Based on the clinical trial research data of my country's approved vaccines and the epidemiological characteristics of new coronary pneumonia, the New Coronavirus Vaccine Working Group of the Chinese Center for Disease Control and Prevention drafted this guide, which was reviewed and approved by the Expert Advisory Committee of the National Immunization Program.
This guide is used by health departments and disease control agencies at all levels to guide vaccination units to carry out new coronavirus vaccination.
1.
Types of vaccines (1) Inactivated vaccines.
The three new coronavirus inactivated vaccine products approved for listing with conditions are provided by Sinopharm China National Bio-Beijing Institute of Biological Products Co.
, Ltd.
(Beijing Institute), Wuhan Institute of Biological Products Co.
, Ltd.
(Wuhan Institute) and Beijing Kexing Zhongwei Produced by Biotechnology Co.
, Ltd.
(Kexing Zhongwei). The principle is to use African green monkey kidney (Vero) cells for virus culture and amplification.
The virus is inactivated by β-propiolactone, antigen components are retained to induce the body to produce an immune response, and aluminum hydroxide adjuvant is added to improve immunogenicity.
.
(2) Adenovirus vector vaccine.
Conditionally approved adenovirus vector vaccine is a recombinant new coronavirus vaccine (type 5 adenovirus vector) produced by Cansino Biotech Co.
, Ltd.
(Cansino).
The principle is to recombine the spike glycoprotein (S protein) gene of the new coronavirus into the replication-deficient human type 5 adenovirus gene.
The genetically recombined adenovirus expresses the new coronavirus S protein antigen in the body and induces the body to produce an immune response.
(3) Recombinant subunit vaccine.
The recombinant subunit vaccine approved for emergency use is the recombinant new coronavirus vaccine (CHO cell) produced by Anhui Zhifeilongkoma Biopharmaceutical Co.
, Ltd.
(Zhifeilongkoma).
The principle is to recombine the new coronavirus S protein receptor binding region (RBD) gene into Chinese hamster ovary (CHO) cell genes, express in vitro to form RBD dimer, and add aluminum hydroxide adjuvant to improve immunogenicity .
2.
Recommended immunization procedures (1) Applicable objects.
People who are 18 years old and above.
(2) Times and intervals of inoculation doses.
1.
Two doses of inactivated new coronavirus vaccine (Vero cells); the recommended interval between the two doses is ≥3 weeks, and the second dose should be completed as soon as possible within 8 weeks.
2.
One dose of recombinant new coronavirus vaccine (type 5 adenovirus vector).
3.
Three doses of recombinant new coronavirus vaccine (CHO cells) are inoculated; the recommended interval between two adjacent doses is ≥4 weeks.
The second dose should be completed within 8 weeks after the first dose, and the third dose should be completed within 6 months after the first dose.
(3) The route of vaccination and the site of vaccination.
Intramuscular injection in the deltoid muscle of the upper arm is recommended.
3.
Other related matters (1) Late planting and replanting. For the two-dose or three-dose program, if the vaccination is not completed according to the program, it is recommended to replant as soon as possible.
The immunization program does not need to be restarted, just complete the corresponding doses.
For those who have completed 2 doses of inactivated new coronavirus vaccination within 14 days, one dose of inactivated vaccine should be supplemented as soon as possible three weeks after the second dose.
For those who have completed 2 doses of inactivated new coronavirus vaccination within 14-21 days, there is no need to replant.
(2) Strengthen immunity.
Boost immunization is not recommended at this stage.
(3) Simultaneous vaccination with other vaccines.
It is not recommended to vaccinate with other vaccines at the same time.
The interval between other vaccines and the new coronavirus vaccine should be greater than 14 days.
When rabies vaccine, tetanus vaccine, and immunoglobulin are needed due to animal injuries, trauma, etc.
, the interval between vaccination with the new coronavirus vaccine may not be considered.
(4) Replacement of different vaccine products.
At this stage, it is recommended to use the same vaccine product to complete the vaccination.
In special circumstances such as the inability to continue the supply of vaccines and the recipients are vaccinated in different places, and the same vaccine product cannot be used to complete the vaccination, the same type of vaccine products from other manufacturers can be used to complete the vaccination.
(5) New coronavirus infection and antibody screening.
There is no need to carry out new coronavirus nucleic acid and antibody testing before vaccination; routine antibody testing is not recommended as a basis for immunization success after vaccination.
(6) Contraindications for vaccination.
The usual contraindications for vaccination include: (1) Those who are allergic to the active ingredient of the vaccine, any inactive ingredient, the substances used in the production process, or those who have had allergies during previous vaccination of the same type of vaccine; (2) Have experienced serious vaccines in the past Allergic reactions (such as acute allergic reactions, angioedema, dyspnea, etc.
); (3) People with uncontrolled epilepsy and other severe neurological diseases (such as transverse myelitis, Guillain-Barre syndrome, demyelination) Sheath diseases, etc.
); (4) Patients with fever, or suffering from acute diseases, or acute attacks of chronic diseases, or patients with uncontrolled severe chronic diseases; (5) Women during pregnancy. This guide will refer to the following vaccination recommendations for specific populations for some people who are contraindicated or used with caution in the instructions.
4.
Recommendations for vaccination for specific populations (1) People aged 60 and above.
People aged 60 and above are those at high risk of severe illness and death after being infected with the new coronavirus.
At present, the number of phase III clinical trials of the 4 new coronavirus vaccines with conditional approval for marketing has been included in this population in a limited number, and there is no data on the protective efficacy of the vaccine in this population.
However, the data of phase I/II clinical studies show that this population has good vaccination safety.
Compared with the population of 18-59 years old, the neutralizing antibody titer after vaccination is slightly lower, but the positive conversion rate of neutralizing antibody is similar, suggesting that the vaccine is 60% People over the age of age will also have a certain protective effect, and vaccination is recommended.
(2) People under 18 years old.
At present, the existing vaccines have not yet obtained clinical trial data for this population, and it is not recommended for people under 18 years of age to be vaccinated.
(3) People with chronic diseases.
People with chronic diseases are those who are severely ill and at high risk of death after being infected with the new coronavirus.
People with stable health conditions and well-controlled chronic diseases are not considered contraindications for new coronavirus vaccination and are recommended to be vaccinated.
(4) Women of childbearing age and breastfeeding period.
If you get pregnant after vaccination or get the vaccine without knowing your pregnancy, based on the understanding of the safety of the above vaccines, it is not recommended to take special medical measures (such as termination of pregnancy) just because of the new coronavirus vaccine.
It is recommended to do a pregnancy check and Follow up.
For women who have a pregnancy plan, there is no need to delay the pregnancy plan just because of the new coronavirus vaccine.
Although there is currently no clinical research data on the impact of new coronavirus vaccines in breastfeeding women on breastfeeding infants, based on the understanding of the safety of the vaccine, it is recommended to vaccinate breastfeeding women (such as medical staff) who are at high risk of new coronavirus infections.
.
Considering the importance of breastfeeding to the nutrition and health of infants and young children, referring to internationally accepted practices, breastfeeding women are recommended to continue breastfeeding after being vaccinated against the new coronavirus.
(5) People with impaired immune function.
People with impaired immune function are those at high risk of severe illness and death after being infected with the new coronavirus.
There is currently no data on the safety and effectiveness of the new coronavirus vaccine for the population (such as patients with malignant tumors, nephrotic syndrome, AIDS) and people infected with human immunodeficiency virus (HIV).
The immune response and protective effect of this group of people after vaccination may be reduced.
For inactivated vaccines and recombinant subunit vaccines, based on the safety characteristics of previous vaccines of the same type, vaccination is recommended; for adenovirus vector vaccines, although the vector virus used is replication defective, there is no safety data for the use of the same type of vaccine in the past.
It is recommended that after being fully informed, the individual weighs the benefits outweigh the risks and vaccinates.
(6) Past patients or infected persons with new coronary disease.
Existing research data shows that there are rare cases of re-infection within 6 months after the new coronavirus infection.
People who have been infected with the new crown pneumonia virus (patients or asymptomatic infections) can receive one dose after 6 months on the basis of full notification.
5.
Other matters As more new coronavirus vaccines are approved for use, vaccine clinical research data continues to improve, and vaccine monitoring and evaluation data increase after the market is launched, this guide will be updated in due course according to the needs of the epidemic prevention and control situation.
The guide mentions that people with chronic diseases who are well controlled by drugs are not considered contraindicated for new coronavirus vaccination and are recommended to be vaccinated.
Source: National Health Commission's official website.
The National Health Commission's website reported on March 29 that at present, my country has 5 manufacturers of new coronavirus vaccines approved for conditional listing or emergency use.
The results of the interim analysis of the phase III clinical trials of the three inactivated vaccines and adenovirus vector vaccines that were conditionally approved for marketing showed that the protective efficacy of the vaccines all met the guidelines for the clinical evaluation of new coronavirus preventive vaccines (trial) by the National Medical Products Administration.
The requirements also meet the index requirements recommended by the World Health Organization’s "New Coronavirus Vaccine Target Product Characteristics".
Surveillance data of suspected abnormal response to vaccination after large-scale vaccination in key populations during clinical trials and emergency use phases and earlier stages show that the new coronavirus vaccine is safe.
The results of the Phase II clinical trial of the recombinant new coronavirus vaccine (CHO cell) approved for emergency use showed good immunogenicity and safety.
Based on the clinical trial research data of my country's approved vaccines and the epidemiological characteristics of new coronary pneumonia, the New Coronavirus Vaccine Working Group of the Chinese Center for Disease Control and Prevention drafted this guide, which was reviewed and approved by the Expert Advisory Committee of the National Immunization Program.
This guide is used by health departments and disease control agencies at all levels to guide vaccination units to carry out new coronavirus vaccination.
1.
Types of vaccines (1) Inactivated vaccines.
The three new coronavirus inactivated vaccine products approved for listing with conditions are provided by Sinopharm China National Bio-Beijing Institute of Biological Products Co.
, Ltd.
(Beijing Institute), Wuhan Institute of Biological Products Co.
, Ltd.
(Wuhan Institute) and Beijing Kexing Zhongwei Produced by Biotechnology Co.
, Ltd.
(Kexing Zhongwei). The principle is to use African green monkey kidney (Vero) cells for virus culture and amplification.
The virus is inactivated by β-propiolactone, antigen components are retained to induce the body to produce an immune response, and aluminum hydroxide adjuvant is added to improve immunogenicity.
.
(2) Adenovirus vector vaccine.
Conditionally approved adenovirus vector vaccine is a recombinant new coronavirus vaccine (type 5 adenovirus vector) produced by Cansino Biotech Co.
, Ltd.
(Cansino).
The principle is to recombine the spike glycoprotein (S protein) gene of the new coronavirus into the replication-deficient human type 5 adenovirus gene.
The genetically recombined adenovirus expresses the new coronavirus S protein antigen in the body and induces the body to produce an immune response.
(3) Recombinant subunit vaccine.
The recombinant subunit vaccine approved for emergency use is the recombinant new coronavirus vaccine (CHO cell) produced by Anhui Zhifeilongkoma Biopharmaceutical Co.
, Ltd.
(Zhifeilongkoma).
The principle is to recombine the new coronavirus S protein receptor binding region (RBD) gene into Chinese hamster ovary (CHO) cell genes, express in vitro to form RBD dimer, and add aluminum hydroxide adjuvant to improve immunogenicity .
2.
Recommended immunization procedures (1) Applicable objects.
People who are 18 years old and above.
(2) Times and intervals of inoculation doses.
1.
Two doses of inactivated new coronavirus vaccine (Vero cells); the recommended interval between the two doses is ≥3 weeks, and the second dose should be completed as soon as possible within 8 weeks.
2.
One dose of recombinant new coronavirus vaccine (type 5 adenovirus vector).
3.
Three doses of recombinant new coronavirus vaccine (CHO cells) are inoculated; the recommended interval between two adjacent doses is ≥4 weeks.
The second dose should be completed within 8 weeks after the first dose, and the third dose should be completed within 6 months after the first dose.
(3) The route of vaccination and the site of vaccination.
Intramuscular injection in the deltoid muscle of the upper arm is recommended.
3.
Other related matters (1) Late planting and replanting. For the two-dose or three-dose program, if the vaccination is not completed according to the program, it is recommended to replant as soon as possible.
The immunization program does not need to be restarted, just complete the corresponding doses.
For those who have completed 2 doses of inactivated new coronavirus vaccination within 14 days, one dose of inactivated vaccine should be supplemented as soon as possible three weeks after the second dose.
For those who have completed 2 doses of inactivated new coronavirus vaccination within 14-21 days, there is no need to replant.
(2) Strengthen immunity.
Boost immunization is not recommended at this stage.
(3) Simultaneous vaccination with other vaccines.
It is not recommended to vaccinate with other vaccines at the same time.
The interval between other vaccines and the new coronavirus vaccine should be greater than 14 days.
When rabies vaccine, tetanus vaccine, and immunoglobulin are needed due to animal injuries, trauma, etc.
, the interval between vaccination with the new coronavirus vaccine may not be considered.
(4) Replacement of different vaccine products.
At this stage, it is recommended to use the same vaccine product to complete the vaccination.
In special circumstances such as the inability to continue the supply of vaccines and the recipients are vaccinated in different places, and the same vaccine product cannot be used to complete the vaccination, the same type of vaccine products from other manufacturers can be used to complete the vaccination.
(5) New coronavirus infection and antibody screening.
There is no need to carry out new coronavirus nucleic acid and antibody testing before vaccination; routine antibody testing is not recommended as a basis for immunization success after vaccination.
(6) Contraindications for vaccination.
The usual contraindications for vaccination include: (1) Those who are allergic to the active ingredient of the vaccine, any inactive ingredient, the substances used in the production process, or those who have had allergies during previous vaccination of the same type of vaccine; (2) Have experienced serious vaccines in the past Allergic reactions (such as acute allergic reactions, angioedema, dyspnea, etc.
); (3) People with uncontrolled epilepsy and other severe neurological diseases (such as transverse myelitis, Guillain-Barre syndrome, demyelination) Sheath diseases, etc.
); (4) Patients with fever, or suffering from acute diseases, or acute attacks of chronic diseases, or patients with uncontrolled severe chronic diseases; (5) Women during pregnancy. This guide will refer to the following vaccination recommendations for specific populations for some people who are contraindicated or used with caution in the instructions.
4.
Recommendations for vaccination for specific populations (1) People aged 60 and above.
People aged 60 and above are those at high risk of severe illness and death after being infected with the new coronavirus.
At present, the number of phase III clinical trials of the 4 new coronavirus vaccines with conditional approval for marketing has been included in this population in a limited number, and there is no data on the protective efficacy of the vaccine in this population.
However, the data of phase I/II clinical studies show that this population has good vaccination safety.
Compared with the population of 18-59 years old, the neutralizing antibody titer after vaccination is slightly lower, but the positive conversion rate of neutralizing antibody is similar, suggesting that the vaccine is 60% People over the age of age will also have a certain protective effect, and vaccination is recommended.
(2) People under 18 years old.
At present, the existing vaccines have not yet obtained clinical trial data for this population, and it is not recommended for people under 18 years of age to be vaccinated.
(3) People with chronic diseases.
People with chronic diseases are those who are severely ill and at high risk of death after being infected with the new coronavirus.
People with stable health conditions and well-controlled chronic diseases are not considered contraindications for new coronavirus vaccination and are recommended to be vaccinated.
(4) Women of childbearing age and breastfeeding period.
If you get pregnant after vaccination or get the vaccine without knowing your pregnancy, based on the understanding of the safety of the above vaccines, it is not recommended to take special medical measures (such as termination of pregnancy) just because of the new coronavirus vaccine.
It is recommended to do a pregnancy check and Follow up.
For women who have a pregnancy plan, there is no need to delay the pregnancy plan just because of the new coronavirus vaccine.
Although there is currently no clinical research data on the impact of new coronavirus vaccines in breastfeeding women on breastfeeding infants, based on the understanding of the safety of the vaccine, it is recommended to vaccinate breastfeeding women (such as medical staff) who are at high risk of new coronavirus infections.
.
Considering the importance of breastfeeding to the nutrition and health of infants and young children, referring to internationally accepted practices, breastfeeding women are recommended to continue breastfeeding after being vaccinated against the new coronavirus.
(5) People with impaired immune function.
People with impaired immune function are those at high risk of severe illness and death after being infected with the new coronavirus.
There is currently no data on the safety and effectiveness of the new coronavirus vaccine for the population (such as patients with malignant tumors, nephrotic syndrome, AIDS) and people infected with human immunodeficiency virus (HIV).
The immune response and protective effect of this group of people after vaccination may be reduced.
For inactivated vaccines and recombinant subunit vaccines, based on the safety characteristics of previous vaccines of the same type, vaccination is recommended; for adenovirus vector vaccines, although the vector virus used is replication defective, there is no safety data for the use of the same type of vaccine in the past.
It is recommended that after being fully informed, the individual weighs the benefits outweigh the risks and vaccinates.
(6) Past patients or infected persons with new coronary disease.
Existing research data shows that there are rare cases of re-infection within 6 months after the new coronavirus infection.
People who have been infected with the new crown pneumonia virus (patients or asymptomatic infections) can receive one dose after 6 months on the basis of full notification.
5.
Other matters As more new coronavirus vaccines are approved for use, vaccine clinical research data continues to improve, and vaccine monitoring and evaluation data increase after the market is launched, this guide will be updated in due course according to the needs of the epidemic prevention and control situation.