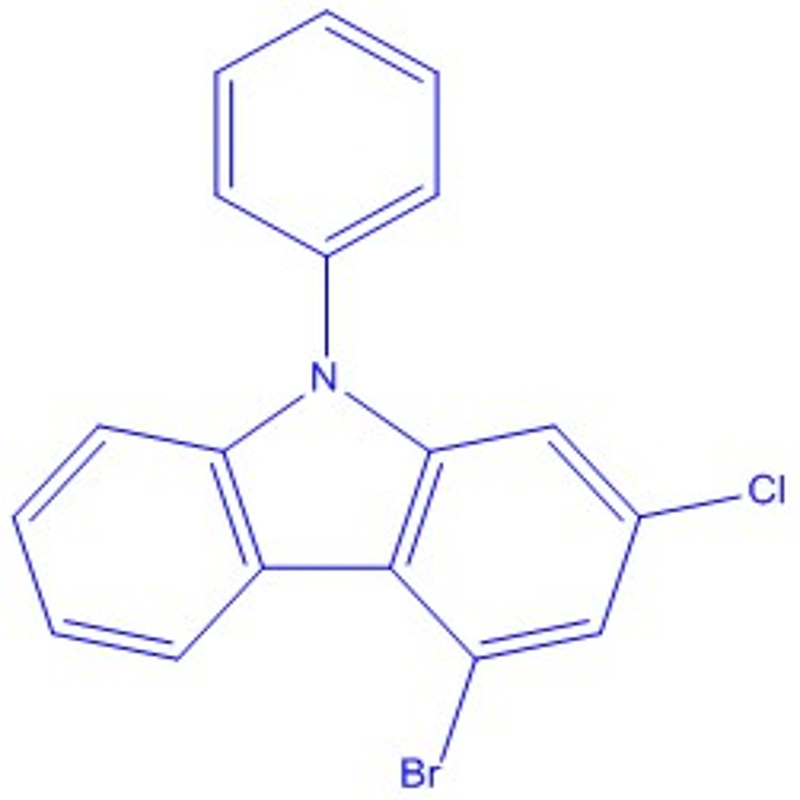
-
Categories
-
Pharmaceutical Intermediates
-
Active Pharmaceutical Ingredients
-
Food Additives
- Industrial Coatings
- Agrochemicals
- Dyes and Pigments
- Surfactant
- Flavors and Fragrances
- Chemical Reagents
- Catalyst and Auxiliary
- Natural Products
- Inorganic Chemistry
-
Organic Chemistry
-
Biochemical Engineering
- Analytical Chemistry
-
Cosmetic Ingredient
- Water Treatment Chemical
-
Pharmaceutical Intermediates
Promotion
ECHEMI Mall
Wholesale
Weekly Price
Exhibition
News
-
Trade Service
Since the release of the new regulations, only a few companies related to China have publicly applied for listing in the United States
.
Recently, the clinical-stage biopharmaceutical company Liantuo Biopharmaceuticals disclosed the prospectus, and the company will be listed on the Nasdaq in the United States on October 28
.
The company issued 20.
3 million American Depositary Shares (ADS) at a price of US$15 to US$17 per share.
Based on the upper limit of the issuance range, the IPO valuation of Liantuo Bio will be approximately US$1.
8 billion
.
A clinical-stage biopharmaceutical company operating in China and the United States.
Founded in 2019, Liantuo Bio is a science-oriented global biopharmaceutical company.
The company is headquartered in Princeton, New Jersey, USA, and mainly conducts business in China
.
The company is committed to developing and commercializing innovative drugs for patients with unmet medical needs, initially focusing on the licensing, development and commercialization of therapeutic drugs in Greater China (including Mainland China, Hong Kong, Taiwan, and Macau) and other Asian markets
.
Currently, the company is developing nine candidate products, of which three are about to enter the third phase of testing
.
Its partners include pharmaceutical giant Bristol-Myers Squibb, Tarsus Pharmaceuticals and Pfizer
.
It is understood that at the beginning of the establishment of LinkBio, BridgeBio Pharma announced a cooperation with LinkBio to expand its pipeline to China
.
The initial cooperation will focus on two oncology products developed by BridgeBio, including the selective FGFR inhibitor infigratinib, which is currently undergoing phase 3 clinical trials, and the SHP2 inhibitor BBP, which is used to treat solid tumors caused by RAS and tyrosine kinase receptors.
-398
.
According to the terms of the agreement, Liantuo Bio will obtain commercialization rights in China and designated Asian markets, and participate in the clinical research and development of infigratinib and BBP-398
.
At the same time, Liantuo Biotech has obtained the priority right to use more than 20 pipeline products of BridgeBio in China and other major Asian markets..
This will accelerate the advancement of BridgeBio's clinical research and commercialization in the region, allowing it to quickly solve the medical needs of a large group of people with innovative methods
.
Among them, the anti-tumor candidate drug infigratinib is currently in the phase 3 development stage for the treatment of diseases such as cholangiocarcinoma (CCA) and urothelial carcinoma (bladder cancer) caused by FGFR gene abnormalities
.
Currently at a loss.
The prospectus shows that Liantuo Bio is currently at a loss
.
In 2019 and 2020, Liantuo had operating losses of US$24.
34 million and US$135 million, respectively; Liantuo had an operating loss of US$160 million in the first half of 2021 and an operating loss of US$6.
62 million in the same period last year
.
However, in terms of research and development, the company is increasing its investment.
In 2020, the research and development expenditure of Liantuo will be 120 million U.
S.
dollars, and the research and development expenditure in the first half of 2021 will be 146 million U.
S.
dollars
.
It is worth mentioning that the company previously completed a cross-round financing of more than 300 million US dollars in November 2020.
Investors including Perceptive Advisors, RA Capital, Venrock Healthcare Capital Partners and SDIC China Merchants participated in the joint participation of Chinese and American investors.
.
Other biopharmaceutical investors also include: funds under BlackRock Assets, Casdin Capital, Farallon, Logos Capital, Perceptive Advisors, Pfizer, Sphera Healthcare, funds under T.
Rowe Price Associates, Inc, Tybourne Capital Management, Vida Ventures, Viking Global Investors and Wellington Management
.
In this prospectus, Liantuo Bio emphasizes that China, as the world’s second largest pharmaceutical market, has an increasingly important strategic position, and Western biopharmaceutical companies still have certain challenges to enter the Chinese market.
Therefore, Liantuo’s vision has become "The entrance of western biopharmaceutical companies into the Chinese market"
.
.
Recently, the clinical-stage biopharmaceutical company Liantuo Biopharmaceuticals disclosed the prospectus, and the company will be listed on the Nasdaq in the United States on October 28
.
The company issued 20.
3 million American Depositary Shares (ADS) at a price of US$15 to US$17 per share.
Based on the upper limit of the issuance range, the IPO valuation of Liantuo Bio will be approximately US$1.
8 billion
.
A clinical-stage biopharmaceutical company operating in China and the United States.
Founded in 2019, Liantuo Bio is a science-oriented global biopharmaceutical company.
The company is headquartered in Princeton, New Jersey, USA, and mainly conducts business in China
.
The company is committed to developing and commercializing innovative drugs for patients with unmet medical needs, initially focusing on the licensing, development and commercialization of therapeutic drugs in Greater China (including Mainland China, Hong Kong, Taiwan, and Macau) and other Asian markets
.
Currently, the company is developing nine candidate products, of which three are about to enter the third phase of testing
.
Its partners include pharmaceutical giant Bristol-Myers Squibb, Tarsus Pharmaceuticals and Pfizer
.
It is understood that at the beginning of the establishment of LinkBio, BridgeBio Pharma announced a cooperation with LinkBio to expand its pipeline to China
.
The initial cooperation will focus on two oncology products developed by BridgeBio, including the selective FGFR inhibitor infigratinib, which is currently undergoing phase 3 clinical trials, and the SHP2 inhibitor BBP, which is used to treat solid tumors caused by RAS and tyrosine kinase receptors.
-398
.
According to the terms of the agreement, Liantuo Bio will obtain commercialization rights in China and designated Asian markets, and participate in the clinical research and development of infigratinib and BBP-398
.
At the same time, Liantuo Biotech has obtained the priority right to use more than 20 pipeline products of BridgeBio in China and other major Asian markets..
This will accelerate the advancement of BridgeBio's clinical research and commercialization in the region, allowing it to quickly solve the medical needs of a large group of people with innovative methods
.
Among them, the anti-tumor candidate drug infigratinib is currently in the phase 3 development stage for the treatment of diseases such as cholangiocarcinoma (CCA) and urothelial carcinoma (bladder cancer) caused by FGFR gene abnormalities
.
Currently at a loss.
The prospectus shows that Liantuo Bio is currently at a loss
.
In 2019 and 2020, Liantuo had operating losses of US$24.
34 million and US$135 million, respectively; Liantuo had an operating loss of US$160 million in the first half of 2021 and an operating loss of US$6.
62 million in the same period last year
.
However, in terms of research and development, the company is increasing its investment.
In 2020, the research and development expenditure of Liantuo will be 120 million U.
S.
dollars, and the research and development expenditure in the first half of 2021 will be 146 million U.
S.
dollars
.
It is worth mentioning that the company previously completed a cross-round financing of more than 300 million US dollars in November 2020.
Investors including Perceptive Advisors, RA Capital, Venrock Healthcare Capital Partners and SDIC China Merchants participated in the joint participation of Chinese and American investors.
.
Other biopharmaceutical investors also include: funds under BlackRock Assets, Casdin Capital, Farallon, Logos Capital, Perceptive Advisors, Pfizer, Sphera Healthcare, funds under T.
Rowe Price Associates, Inc, Tybourne Capital Management, Vida Ventures, Viking Global Investors and Wellington Management
.
In this prospectus, Liantuo Bio emphasizes that China, as the world’s second largest pharmaceutical market, has an increasingly important strategic position, and Western biopharmaceutical companies still have certain challenges to enter the Chinese market.
Therefore, Liantuo’s vision has become "The entrance of western biopharmaceutical companies into the Chinese market"
.