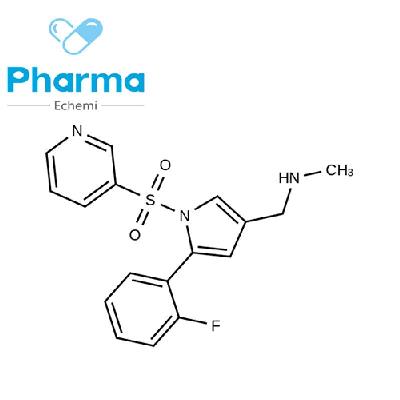
-
Categories
-
Pharmaceutical Intermediates
-
Active Pharmaceutical Ingredients
-
Food Additives
- Industrial Coatings
- Agrochemicals
- Dyes and Pigments
- Surfactant
- Flavors and Fragrances
- Chemical Reagents
- Catalyst and Auxiliary
- Natural Products
- Inorganic Chemistry
-
Organic Chemistry
-
Biochemical Engineering
- Analytical Chemistry
-
Cosmetic Ingredient
- Water Treatment Chemical
-
Pharmaceutical Intermediates
Promotion
ECHEMI Mall
Wholesale
Weekly Price
Exhibition
News
-
Trade Service
▎The content team editor of WuXi AppTec recently, Merus announced the latest clinical trial results of its in-development bispecific antibody therapy zenocutuzumab at the ASCO annual meeting
.
In the treatment of pancreatic cancer patients with NRG1 fusion-positive, zenocutuzumab achieved a confirmed total remission rate of 42% (5/12)
.
When treating NRG1 fusion-positive non-small cell lung cancer patients, zenocutuzumab achieved an ORR of 25%
.
Zenocutuzumab is a bispecific antibody that targets both HER2 and HER3 receptors, designed to accurately treat patients with solid tumors that are positive for NRG1 fusion
.
In patients with NRG1 fusion-positive tumors, the NRG1 gene that expresses neuregulin is fused with other genes, resulting in abnormal expression of neuregulin, which promotes the occurrence and proliferation of cancer
.
Zenocutuzumab is a bispecific antibody that can simultaneously bind to HER2 and HER3 receptors.
It can prevent HER2 and HER3 from forming dimers, thereby blocking the signal transduction of neuregulin
.
In addition, this antibody also has the effect of enhancing antibody-dependent cell-mediated cytotoxicity (ADCC), thereby improving the killing of tumors by immune cells
.
▲The mechanism of action of Zenocutuzumab (picture source: Merus official website) The results reported by ASCO included 45 patients with NRG1 fusion-positive solid tumors whose results can be evaluated.
The study was conducted in 4 types of cancer patients with multiple different types of NRG1 fusion mutations.
Personnel observed partial relief
.
As of April 13, the total remission rate of 45 patients was 29%, and 40% of evaluable patients were still receiving treatment, and the duration of remission ranged from 1 month to 12 months
.
The lead author of this study, Dr.
Alison Schram of Memorial Sloan Kettering Cancer Center (MSKCC), said: "We are very excited to see the emergence of potential innovative treatments for patients with NRG1 fusion-positive cancers
.
These patients have unmet needs
.
Today .
The published data is the first clinical verification that NRG1 fusion is the driving factor of cancer, and zenocutuzumab can block tumor growth in patients with these fusion variants
.
"Reference: [1] Merus Presents Clinical Data on Zenocutuzumab in NRG1-fusion (NRG1+ ) Cancers at the American Society of Clinical Oncology (ASCO) 2021 Annual Meeting (Oral Abstract).
Retrieved June 4, 2021, from https://ir.
merus.
nl/news-releases/news-release-details/merus-presents -clinical-data-zenocutuzumab-nrg1-fusion-nrg1 Note: This article aims to introduce the progress of medical and health research, not a treatment plan recommendation
.
If you need guidance on the treatment plan, please go to a regular hospital for treatment
.
.
In the treatment of pancreatic cancer patients with NRG1 fusion-positive, zenocutuzumab achieved a confirmed total remission rate of 42% (5/12)
.
When treating NRG1 fusion-positive non-small cell lung cancer patients, zenocutuzumab achieved an ORR of 25%
.
Zenocutuzumab is a bispecific antibody that targets both HER2 and HER3 receptors, designed to accurately treat patients with solid tumors that are positive for NRG1 fusion
.
In patients with NRG1 fusion-positive tumors, the NRG1 gene that expresses neuregulin is fused with other genes, resulting in abnormal expression of neuregulin, which promotes the occurrence and proliferation of cancer
.
Zenocutuzumab is a bispecific antibody that can simultaneously bind to HER2 and HER3 receptors.
It can prevent HER2 and HER3 from forming dimers, thereby blocking the signal transduction of neuregulin
.
In addition, this antibody also has the effect of enhancing antibody-dependent cell-mediated cytotoxicity (ADCC), thereby improving the killing of tumors by immune cells
.
▲The mechanism of action of Zenocutuzumab (picture source: Merus official website) The results reported by ASCO included 45 patients with NRG1 fusion-positive solid tumors whose results can be evaluated.
The study was conducted in 4 types of cancer patients with multiple different types of NRG1 fusion mutations.
Personnel observed partial relief
.
As of April 13, the total remission rate of 45 patients was 29%, and 40% of evaluable patients were still receiving treatment, and the duration of remission ranged from 1 month to 12 months
.
The lead author of this study, Dr.
Alison Schram of Memorial Sloan Kettering Cancer Center (MSKCC), said: "We are very excited to see the emergence of potential innovative treatments for patients with NRG1 fusion-positive cancers
.
These patients have unmet needs
.
Today .
The published data is the first clinical verification that NRG1 fusion is the driving factor of cancer, and zenocutuzumab can block tumor growth in patients with these fusion variants
.
"Reference: [1] Merus Presents Clinical Data on Zenocutuzumab in NRG1-fusion (NRG1+ ) Cancers at the American Society of Clinical Oncology (ASCO) 2021 Annual Meeting (Oral Abstract).
Retrieved June 4, 2021, from https://ir.
merus.
nl/news-releases/news-release-details/merus-presents -clinical-data-zenocutuzumab-nrg1-fusion-nrg1 Note: This article aims to introduce the progress of medical and health research, not a treatment plan recommendation
.
If you need guidance on the treatment plan, please go to a regular hospital for treatment
.