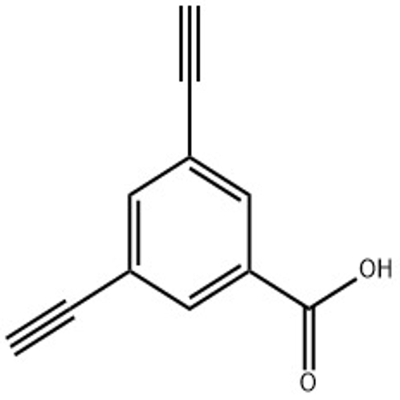
-
Categories
-
Pharmaceutical Intermediates
-
Active Pharmaceutical Ingredients
-
Food Additives
- Industrial Coatings
- Agrochemicals
- Dyes and Pigments
- Surfactant
- Flavors and Fragrances
- Chemical Reagents
- Catalyst and Auxiliary
- Natural Products
- Inorganic Chemistry
-
Organic Chemistry
-
Biochemical Engineering
- Analytical Chemistry
-
Cosmetic Ingredient
- Water Treatment Chemical
-
Pharmaceutical Intermediates
Promotion
ECHEMI Mall
Wholesale
Weekly Price
Exhibition
News
-
Trade Service
Medical Network News, August 3, a new round of medical insurance negotiations officially opened, the adjustment work will be completed before the end of this year, and strive to implement it in January next year
.
At present, the second phase of the application has been completed, and 271 drugs have passed the review
.
On July 30, the State Administration of Medicine issued the "Announcement on Publicizing Drugs and Information that Passed the Preliminary Formal Examination of the 2021 National Medical Insurance Drug Catalogue Adjustment", and 271 medicines passed the preliminary form examination (see the attached table at the end of the article for details)
.
Among them:
Western medicines and proprietary Chinese medicines out of the catalogue:
Condition 1: New generic drugs approved for marketing by the State Drug Administration during the period from January 1, 2016 to June 30, 2021
.
160 varieties involved.
Condition 2: During the period from January 1, 2016 to June 30, 2021, drugs with major changes in indications or functions and indications approved by the State Drug Administration
.
13 varieties are involved.
Condition 4: Drugs included in the National Essential Drug List (2018 Edition)
.
Involving 6 typesof Western medicines and proprietary Chinese medicines in the
catalogue:
Condition 1: An exclusive negotiated medicine that will expire on December 31, 2021, and the payment standard needs to be re-determined in accordance with the agreement
.
74 varieties involved.
Condition 2: During the period from January 1, 2016 to June 30, 2021, with the approval of the State Drug Administration, the indications or functions and indications have undergone major changes and the company actively declares and adjusts the scope of payment for drugs
.
Involving 25 varieties.
Prior to this (June 30), the National Medical Insurance Administration issued the announcement of the "2021 National Medical Insurance Drug List Adjustment Work Plan" and "2021 National Medical Insurance Drug List Adjustment Application Guidelines"
.
The announcement of the plan marked the official start of a new round of adjustments to the national medical insurance drug catalogue
.
At present, after three rounds of adjustments, 433 new drugs have been added to the catalog
.
At the same time, the National Medical Insurance Bureau also interpreted the publicity work of the adjustment of the medical insurance catalogue
.
1.
Does passing the preliminary formal review mean that it has entered the National Medical Insurance Drug List?
A drug that has passed the preliminary formal review does not mean that it has entered the medical insurance catalog.
It only means that the drug has been reviewed and meets the corresponding application conditions and has initially obtained the qualification to participate in the next review
.
According to the "Work Plan for the Adjustment of the National Medical Insurance Drug Catalogue in 2021" (referred to as the "Work Plan"), the catalog adjustment includes multiple stages such as preparation, declaration, review, negotiation, and announcement of results
.
The formal review is only one link in the application stage.
After a drug passes the formal review, it will go through many links.
Only after successfully passing all the links in the adjustment work can it finally enter the national medical insurance catalogue
.
2.
What is the purpose of publicizing the drugs and materials that have passed the preliminary formal review? Publicity is a necessary part of formal review, and it is also an important measure to actively accept social supervision and improve the transparency of catalog adjustment work
.
Since the establishment of the National Medical Insurance Administration, we have been adhering to the direction of openness, fairness and justice, focusing on deepening the reform of the medical insurance management system, establishing and improving a dynamic adjustment mechanism, and continuously optimizing and adjusting procedures and rules.
The level of scientific, standardized, and legalized catalog adjustments is remarkable.
Improvement, and related work has achieved obvious results
.
In accordance with the requirements of the "Work Plan", the declared drugs will be reviewed in terms of effectiveness, safety, economy, innovation, and fairness during the catalog adjustment process
.
In the application process, each application subject submitted the review materials as required, which served as an important reference for expert review
.
In order to ensure that the relevant drugs meet the application conditions and the relevant information is true, accurate and complete, we publicize the drugs and related information on the basis of the company's commitments, and accept supervision from all walks of life
.
Please understand and support the publicity work, fair, objective, comprehensive and accurate interpretation, and all parties are welcome to put forward reasonable opinions and suggestions
.
3.
What are the steps involved in formal review?
Since the implementation of the declaration system, the National Medical Insurance Administration has clarified the declaration conditions according to the "Work Plan" of the year, and each declaration subject voluntarily declares according to the conditions
.
In order to ensure that all declared drugs meet the conditions, it is necessary to conduct a formal review of the drug declaration materials
.
The formal review mainly includes the review of the completeness and standardization of the application materials.
At the same time, we will also verify the authenticity of some materials with relevant parties according to the needs and arrangements of the work
.
This public announcement is one of the links in the formal review
.
4.
What is the basic situation of the review of the adjustment form of the National Medical Insurance Drug List in 2021? From 9:00 on July 1, 2021 to 17:00 on July 14, 2021, a total of 501 pieces of enterprise application information were received, involving 474 drugs
.
After review, 271 drugs passed the preliminary formal review
.
Among the non-catalog drugs that passed the preliminary formal review, the newly marketed drugs after 2016 accounted for 93.
02%
.
5.
How do you think that some of the more expensive drugs currently on the market have passed the formal review? In the previous adjustments to the drug catalogues, the National Medical Insurance Administration resolutely implemented the decisions and deployments of the Party Central Committee and the State Council, always firmly grasped the functional positioning of basic medical insurance "basic", and did its best and did its best to ensure that the newly added drugs in the catalog complied with "Basic" positioning
.
Always take the medical insurance fund and the affordability of the majority of insured persons as the basis for carrying out catalogue adjustments, drastically reduce the price of exclusive drugs through access negotiations , improve accessibility, and maintain fairness
.
Some more expensive drugs have passed the preliminary formal review, which only means that the drug meets the application requirements after the preliminary review and has obtained the qualification to enter the next adjustment link
.
Whether such drugs can eventually enter the National Medical Insurance Drug List must be subject to rigorous review including economics.
Exclusive drugs must undergo price negotiations, and only those who have successfully negotiated can enter the list
.
6.
After the publicity is over, what are the next steps? After the publicity is over, we will process the feedback and suggestions received during the publicity in accordance with the procedures, and determine the scope of drugs that will eventually pass the formal review
.
At the same time, in accordance with the requirements of the "Work Plan", organize and carry out follow-up work such as expert review
.
.
At present, the second phase of the application has been completed, and 271 drugs have passed the review
.
On July 30, the State Administration of Medicine issued the "Announcement on Publicizing Drugs and Information that Passed the Preliminary Formal Examination of the 2021 National Medical Insurance Drug Catalogue Adjustment", and 271 medicines passed the preliminary form examination (see the attached table at the end of the article for details)
.
Among them:
Western medicines and proprietary Chinese medicines out of the catalogue:
Condition 1: New generic drugs approved for marketing by the State Drug Administration during the period from January 1, 2016 to June 30, 2021
.
160 varieties involved.
Condition 2: During the period from January 1, 2016 to June 30, 2021, drugs with major changes in indications or functions and indications approved by the State Drug Administration
.
13 varieties are involved.
Condition 4: Drugs included in the National Essential Drug List (2018 Edition)
.
Involving 6 typesof Western medicines and proprietary Chinese medicines in the
catalogue:
Condition 1: An exclusive negotiated medicine that will expire on December 31, 2021, and the payment standard needs to be re-determined in accordance with the agreement
.
74 varieties involved.
Condition 2: During the period from January 1, 2016 to June 30, 2021, with the approval of the State Drug Administration, the indications or functions and indications have undergone major changes and the company actively declares and adjusts the scope of payment for drugs
.
Involving 25 varieties.
Prior to this (June 30), the National Medical Insurance Administration issued the announcement of the "2021 National Medical Insurance Drug List Adjustment Work Plan" and "2021 National Medical Insurance Drug List Adjustment Application Guidelines"
.
The announcement of the plan marked the official start of a new round of adjustments to the national medical insurance drug catalogue
.
At present, after three rounds of adjustments, 433 new drugs have been added to the catalog
.
At the same time, the National Medical Insurance Bureau also interpreted the publicity work of the adjustment of the medical insurance catalogue
.
1.
Does passing the preliminary formal review mean that it has entered the National Medical Insurance Drug List?
A drug that has passed the preliminary formal review does not mean that it has entered the medical insurance catalog.
It only means that the drug has been reviewed and meets the corresponding application conditions and has initially obtained the qualification to participate in the next review
.
According to the "Work Plan for the Adjustment of the National Medical Insurance Drug Catalogue in 2021" (referred to as the "Work Plan"), the catalog adjustment includes multiple stages such as preparation, declaration, review, negotiation, and announcement of results
.
The formal review is only one link in the application stage.
After a drug passes the formal review, it will go through many links.
Only after successfully passing all the links in the adjustment work can it finally enter the national medical insurance catalogue
.
2.
What is the purpose of publicizing the drugs and materials that have passed the preliminary formal review? Publicity is a necessary part of formal review, and it is also an important measure to actively accept social supervision and improve the transparency of catalog adjustment work
.
Since the establishment of the National Medical Insurance Administration, we have been adhering to the direction of openness, fairness and justice, focusing on deepening the reform of the medical insurance management system, establishing and improving a dynamic adjustment mechanism, and continuously optimizing and adjusting procedures and rules.
The level of scientific, standardized, and legalized catalog adjustments is remarkable.
Improvement, and related work has achieved obvious results
.
In accordance with the requirements of the "Work Plan", the declared drugs will be reviewed in terms of effectiveness, safety, economy, innovation, and fairness during the catalog adjustment process
.
In the application process, each application subject submitted the review materials as required, which served as an important reference for expert review
.
In order to ensure that the relevant drugs meet the application conditions and the relevant information is true, accurate and complete, we publicize the drugs and related information on the basis of the company's commitments, and accept supervision from all walks of life
.
Please understand and support the publicity work, fair, objective, comprehensive and accurate interpretation, and all parties are welcome to put forward reasonable opinions and suggestions
.
3.
What are the steps involved in formal review?
Since the implementation of the declaration system, the National Medical Insurance Administration has clarified the declaration conditions according to the "Work Plan" of the year, and each declaration subject voluntarily declares according to the conditions
.
In order to ensure that all declared drugs meet the conditions, it is necessary to conduct a formal review of the drug declaration materials
.
The formal review mainly includes the review of the completeness and standardization of the application materials.
At the same time, we will also verify the authenticity of some materials with relevant parties according to the needs and arrangements of the work
.
This public announcement is one of the links in the formal review
.
4.
What is the basic situation of the review of the adjustment form of the National Medical Insurance Drug List in 2021? From 9:00 on July 1, 2021 to 17:00 on July 14, 2021, a total of 501 pieces of enterprise application information were received, involving 474 drugs
.
After review, 271 drugs passed the preliminary formal review
.
Among the non-catalog drugs that passed the preliminary formal review, the newly marketed drugs after 2016 accounted for 93.
02%
.
5.
How do you think that some of the more expensive drugs currently on the market have passed the formal review? In the previous adjustments to the drug catalogues, the National Medical Insurance Administration resolutely implemented the decisions and deployments of the Party Central Committee and the State Council, always firmly grasped the functional positioning of basic medical insurance "basic", and did its best and did its best to ensure that the newly added drugs in the catalog complied with "Basic" positioning
.
Always take the medical insurance fund and the affordability of the majority of insured persons as the basis for carrying out catalogue adjustments, drastically reduce the price of exclusive drugs through access negotiations , improve accessibility, and maintain fairness
.
Some more expensive drugs have passed the preliminary formal review, which only means that the drug meets the application requirements after the preliminary review and has obtained the qualification to enter the next adjustment link
.
Whether such drugs can eventually enter the National Medical Insurance Drug List must be subject to rigorous review including economics.
Exclusive drugs must undergo price negotiations, and only those who have successfully negotiated can enter the list
.
6.
After the publicity is over, what are the next steps? After the publicity is over, we will process the feedback and suggestions received during the publicity in accordance with the procedures, and determine the scope of drugs that will eventually pass the formal review
.
At the same time, in accordance with the requirements of the "Work Plan", organize and carry out follow-up work such as expert review
.